Viral Transport Media (VTM) Filters
Whether sterilizing individual ingredients, bulk VTM, or performing a final fill into vials, our PPS filters will prevent unwanted bacteria contamination in your process, ensuring the quality and sterility of your finished VTM product.
Sterile Filtration of COVID-19 Testing Media
Ensure a successful sterility check and avoid disposing of a valuable batch of media with Critical Process Filtration's PPS sterilizing filters.
The PPS filter can be employed in your process by CDC protocols for sterile filtration of VTM.
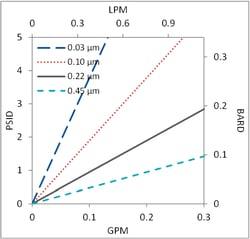
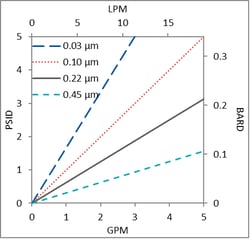
The PPS family of pharmaceutical grade, Polyethersulfone (PES) sterilizing products are constructed with a double layer of PES membrane for greater retention while maintaining high flow and throughput. Available in cartridge, capsule, and mini-capsule devices. Pore sizes recommended for VTM are 0.10 µm and 0.22µm.
PPS Sterilizing Filters
For the most stringent and critical applications.
Applications
- VTM
- Parenterals
- Formulation & Fill
- Diagnostics
- Cell culture media
- Buffers
- Sera/plasma
- Vaccines
- Biologicals
- Ophthalmics
- WFI and Bulk Water
Features and Benefits
/Media%20for%20Product%20Table/PES%20Asymmetric.jpg?width=291&height=200&name=PES%20Asymmetric.jpg)
PPS is a family of pharmaceutical-grade, Polyethersulfone (PES) sterilizing filters. This double-layered, hydrophilic filter is optimized for retention and provides added security when compared to a single-layer model.
- Designed to perform in the most critical applications,
- Delivers high flow and throughput across a wide pH range
- Low binding characteristics (much lower than cellulose acetate and nylon) for filtering liquids with preservatives and proteins that can adsorb to media
- Pore sizes: 0.03 - 1.2 microns
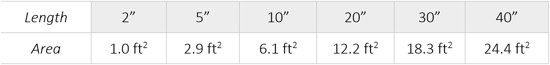
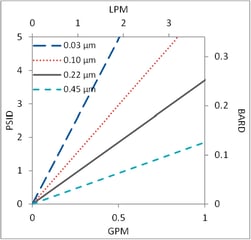
Filtration Area
View the Data Sheet for further product information:
- Integrity test data
- Flow rates of all pore sizes
- Materials of construction
- Operating parameters
- Sanitization and sterilization
- Regulatory & compliance
Validation
PPS filters are validated using test procedures that comply with ASTM F 838-15(ae1) protocols for the determination of bacterial retention in filters used for liquid filtration. The challenge level is a minimum of 107 organisms per cm2 of filter media. CPF filters have > 7-log removal when challenged with the organisms listed below (0.03μm, 0.10μm, and 0.22μm meet the FDA definition of sterilizing grade filters). Validation Guides are available upon request.
- 0.03μm: Acholeplasma laidlawii
- 0.10μm: Brevundimonas diminuta
- 0.22μm: Brevundimonas diminuta
- 0.45μm: Serratia marcescens
- 0.65μm: Saccharomyces cerevisiae
Quality Assurance
PPS filters are 100% integrity tested. They are flushed to remove manufacturing debris and reduce extractability. CPF filters are developed and manufactured under an ISO 9001:2015 certified Quality Management System and designed to be used in cGMP-compliant processes.
PPS Cartridges
Available in multiple sizes from 5-40 inches in length. Several end connections and O-rings options. For applications using elevated temperatures, PPS/HT Cartridges can be implemented up to 203°F.

PPS Capsules
These filters are PPS cartridges encapsulated in a polypropylene shell to minimize exposure to process fluids. A popular choice for single-use applications and can be disposed of easily. Sizes range from 2-30 inches in length. PPS Capsules are available pre-sterilized.

PPS Mini-Capsule
Mini capsules are designed for lab testing and process development. Their overall length with inlet and outlet options ranges from 3.75 - 5.19 inches. PPS Mini-Capsules are available pre-sterilized.