Critical Insights Blog
Are You Using the Right Filters? Bioburden Reduction vs. Sterilizing Filtration
Are You Using the Right Filter? Bioburden Reduction vs. Sterile Filtration Biopharmaceutical processes necessitate stringent microbial control to guarantee product quality and safety. These processes must be carefully designed and operated to
Read MoreAre Sterilizing Filters Reusable?
Are Sterilizing Filters Reusable? It depends. However, is this truly what you need? Furthermore, what is the rationale behind it? While a study demonstrating the filter membrane's capability to sterilize a drug product or process fluid during a
Read More0.22 vs 0.2 Micron Rating -- Is There Really A Difference?
In the realm of sterilizing filtration, micron ratings can be a bit confusing. Some suppliers call out a rating of 0.22µm. Others use 0.2µm. Some actually use both (usually but not always for different products). Is there really a difference?
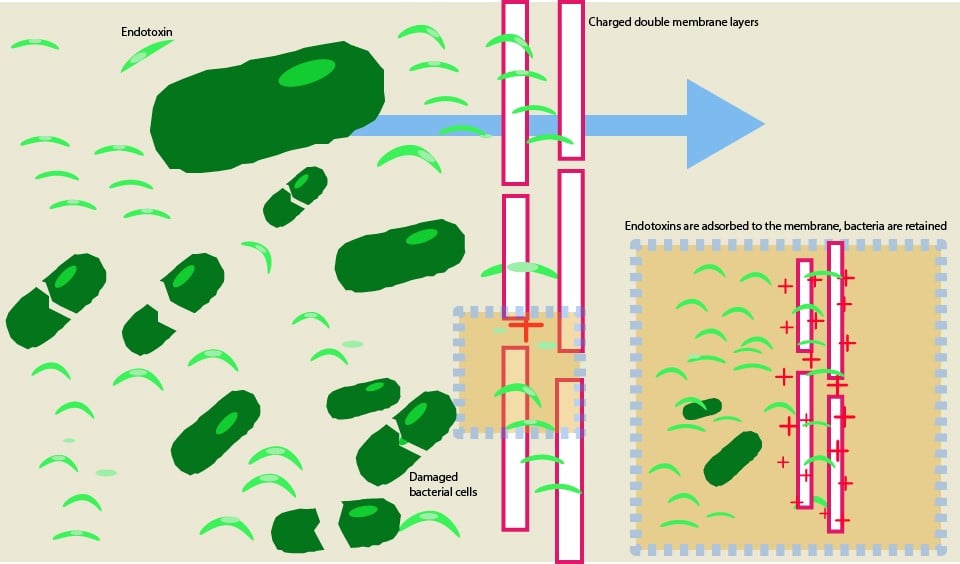
Read MoreThe Difference Between Sterilizing Fluids and Endotoxin Removal
You’ve sanitized your system. You have sterilizing grade filters installed at key points in your process. Your bioburden measurements are low, but you still have endotoxin issues. What’s going on?
Read More