Extractables & Leachables
Extractables & Leachables
Controlling extractables and leachables is an essential aspect of the pharmaceutical manufacturing process. These terms are often misunderstood, necessitating further exploration of their definitions and applications.
Definitions of extractables and leachables tend to overlap and serve distinct purposes. Various manufacturing scenarios must be categorized to emphasize the significance and necessity of both extractables and leachables.
According to the FDA:
- Extractables are compounds that can be extracted from a device (in this case, a filter) when in the presence of a solvent.
- Leachables, on the other hand, are compounds that leach into the drug/product formulation from the device (in this case, a filter) as a result of direct contact with the formulation.
Extractables
Extractables are chemical compounds that can be released from our filters when subjected to rigorous extraction conditions. These compounds are typically identified by exposing the filter to various solvents with different polarities at elevated temperatures. Extractables tests are designed to obtain a fingerprint of different chemicals that can be extracted using standardized conditions. The toxicological review conducted after obtaining extractables is crucial for assessing the product’s risk and selecting the appropriate filter for the intended task.
The primary objective of extractable testing is to validate filter components, screen for and monitor the presence of toxic materials, develop analytical methods for leachable testing, and ensure quality control for the acceptance of filter components.
At Critical Process Filtration, we have outsourced valuation testing on our products for extractables testing. This gives an advantage to our customers when it comes to performing the leachable testing under normal conditions and use of filters.
Leachables
Leachables are chemical compounds that are released by filters into the process solutions under standard operating conditions. These leachables may eventually end up in the final product. Leachable studies can provide information about potential alterations to the product.
Leachable testing monitors and helps control impurities released from filters into the drug products (DPs) during stable production operations or as part of filter qualification studies. Leachables are a subgroup of extractables and can include many of the same compounds that extractables contain, as shown in Figure 1.
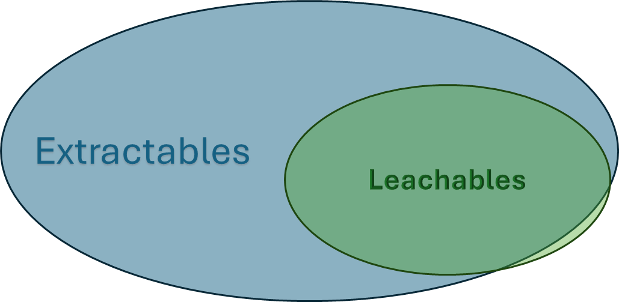
Figure 1. Leachables are a subset of extractables. Extractables testing reveals potential impacts on the product, while leachables identify actual effects.
Compounds Found in Extractables & Leachables
The following table outlines potential extractables associated with various types of membrane filters.
|
Membrane |
Primary Feature |
Extractables Profile |
Major Extractables |
Suggested Filter Application |
|
PES |
High Flow / Low Binding |
Very Low |
Oligomers, Wetting agents |
Biologicals, General Filtration |
|
PVDF |
Low Binding |
Low |
Low molecular weight oligomers, Fluoropolymers |
Proteins, HPLC, Biotech |
|
PTFE |
Chemical Compatibility |
Very Low |
Surfactants for hydrophilic PTFE, Fluorinated residues |
Aggressive Solvents, Vents |
|
Nylon |
High Strength / Hydrophilic |
Low |
Oligomers |
General HPLC, Aqueous Solutions |
Membrane filters are commonly used for sterilizing liquids. Extractables and leachables are a crucial part of a material validation program. Extractables can come from materials like polypropylene and membrane components used during manufacturing. These compounds, including alkanes, oligomers, and wetting agents, can usually be removed from filters through thorough rinsing. This process is essential to prevent these substances from migrating from the filter materials into the final product. Key standards and regulations include:
|
Document |
Requirement |
|
PDA Technical Report No. 26. Comprehensive guidelines for validating extractables and leachables in liquid sterilization filtration. |
Quantifying and identifying extractable substances. Conduct leachables studies whenever feasible. |
|
USP <665>, Plastic Components and Systems Used to Manufacture Pharmaceutical Drug. |
Establishes the necessary criteria for assessing plastic components—like filters, tubing, and bags—that are used in the manufacturing of drug substances and products. |
|
Guidance for Industry for the Submission Documentation for Sterilization Process Validation in Applications for Human and Veterinary Drug Products (FDA). |
Submit extractables information as part of an evaluation of the impact of the filter on the product. |
|
BioPhorum. Best practices guide for evaluating leachables risk from polymeric single-use systems used in biopharmaceutical manufacturing. |
It covers process risk assessment, appropriate solvent extraction procedures, and analytical testing for potential leachables that could impact patient safety. |
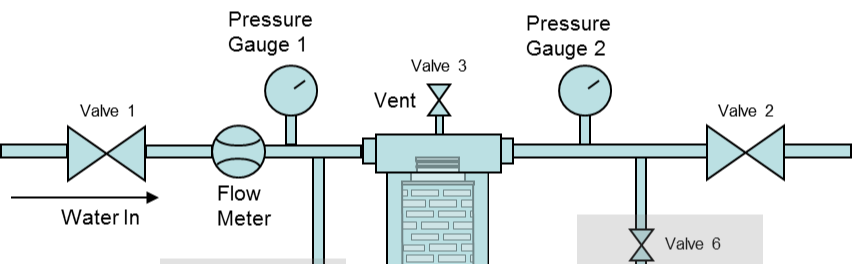
Filter Risk Assessment
The BPOG (Biophorum Operations Group) recommends adopting a risk assessment approach to determine the appropriate level of testing and analysis, taking into account the potential risk posed by leachables from a Single-Use component to the final drug product (Figure 2). It is crucial to consider the following factors:
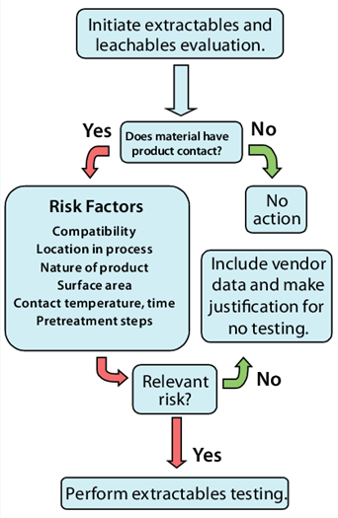
Figure 2. Decision tree for risk analysis of extractables and leachables; W. Ding and M. Jerold, “Implementing Single-Use Technology in Biopharmaceutical Manufacturing,” BioProcess International, 2008.
In the design and execution of leachables testing, it is essential to closely mirror actual process conditions:
|
Aspect |
Details |
|
Process Conditions |
Closely mirror actual process conditions |
|
Duration of Testing |
Should align with real process timeline; leachable concentrations may fluctuate over time |
|
Test Temperatures |
Must reflect maximum process temperature to ensure identification of maximum leachable levels |
|
Pretreatment Steps |
Such as sterilization or rinsing should be carefully replicated during testing due to significant effects on leachables |
Technical Service Collaboration
The Technical Service Team collaborates with customers to develop test plans and conduct tests that closely mimic real-world operating conditions. This approach applies to all application tests, irrespective of the industry, but it’s a prerequisite for validating pharmaceutical processes. These tests are conducted at a laboratory scale using either flat disc filters or encapsulated disc filters.
If you have any further inquiries about our filters or require assistance in determining test values or obtaining additional information, please contact Critical Process Filtration or reach out to us at (603) 880-4420.
Critical Process Filtration, Inc., an ISO 9001 certified manufacturer of process filters, has been assisting customers for over two decades in the design and enhancement of process filtration systems. Our proven products, technical services, support, expedited shipping, and highly competitive pricing ensure that companies receive the best value and expertise. Our comprehensive testing, analysis, and validation support your team whenever they need it. By partnering with your process team, we deliver your company’s solutions effectively and efficiently.