Critical Insights Blog
Troubleshooting Integrity Testing
Troubleshooting Integrity Testing Filter integrity testing is a crucial component of maintaining a filtration system. This is particularly important for sterilizing filters used in biopharmaceutical applications. Integrity testing, conducted both
Read MoreWater Intrusion Integrity Testing - Why and How
Water Intrusion Integrity Testing - Why and How Integrity testing is a vital step in ensuring a filter will perform as designed. This is especially important for sterilizing grade filters, which have been validated to demonstrate performance versus
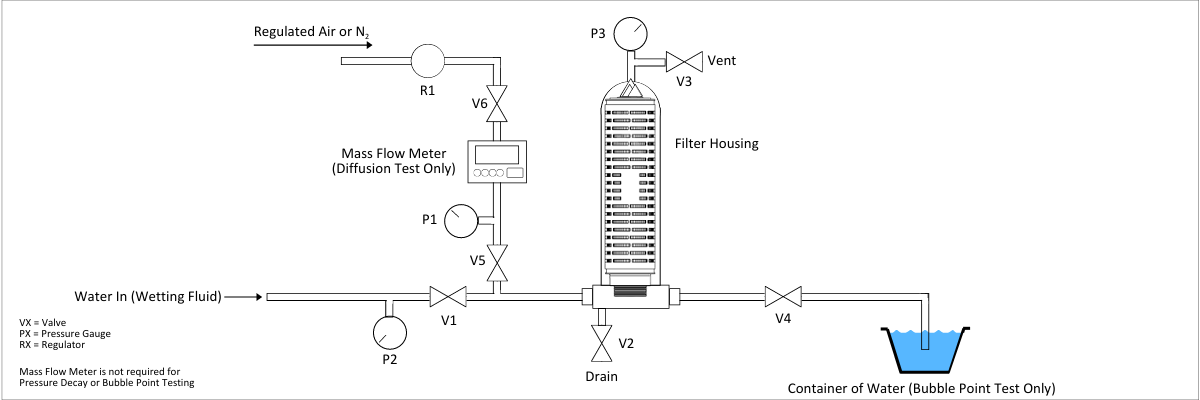
Read MoreIntegrity Testing of Hydrophilic Membrane Filters: Diffusive Flow Test
Preface All integrity test procedures outlined below should be followed when performing integrity testing of membrane filters only. Use of these procedures on other types of filters such as polypropylene or glass fiber cartridges will not give
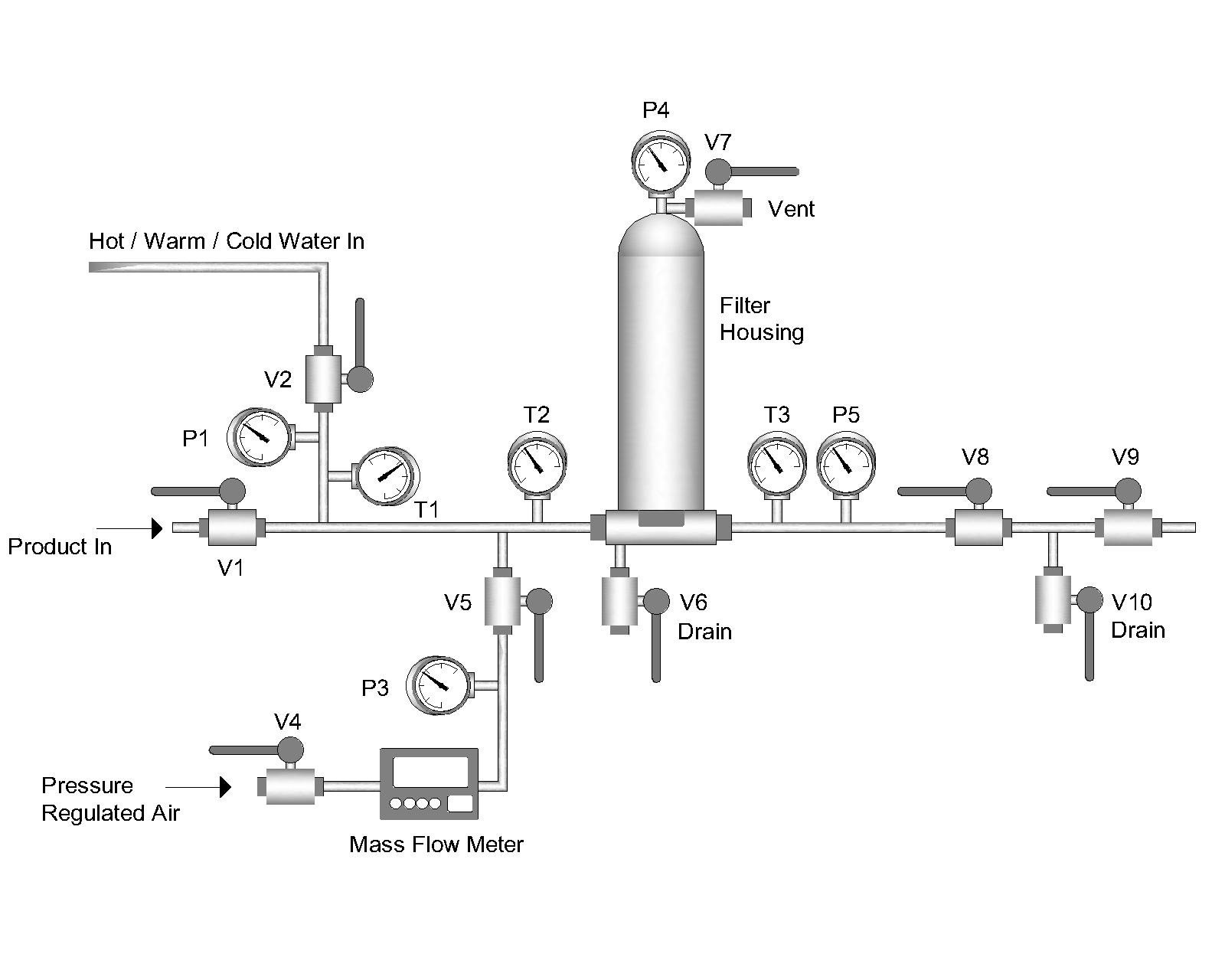
Read MoreFilter Integrity Testing without Capital Expenditure
If you’re using filtration to remove bacteria from your process, integrity testing will provide you with data to ensure product quality. Did you know you can perform integrity testing in-house without the huge expense of buying commercial equipment?
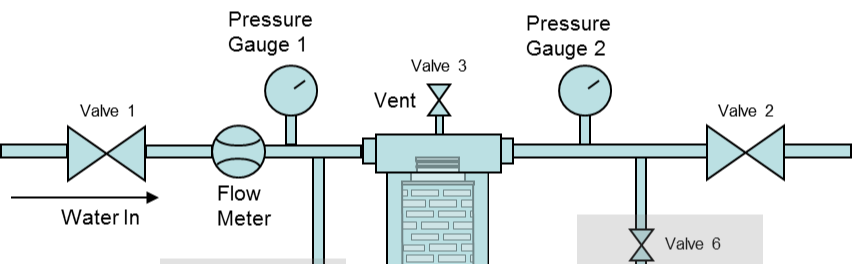
Read MorePressure Decay Testing. Why, When, and How?
A simpler integrity test for less critical applications is a Pressure Decay Test. This requires the same air (or nitrogen) source and an accurate pressure gauge but does not require a mass flow meter. This type of test can be performed before and
Read More