In-Process Filter Integrity Testing. Why, When and How?
Integrity Testing is a requirement of suppliers selling absolute-rated membrane cartridge filters for microbial reduction or removal of yeast and mold. Test results are validated to remove specific organisms, and a certificate is included with each product proving the quality of the filter. In addition to vendor testing, manufacturers in different industries perform their own Integrity Tests for critical processes. In pharmaceutical and bioprocessing, industry regulations call for Integrity Testing at prescribed intervals in a process. In the Food and beverage industry, although not mandatory, Integrity Testing is a strategic move to ensure product safety and quality. Today, we'll discuss the need and steps for in-process integrity testing.
Why test the integrity of a filter that’s already been validated?
Simply put, assurance and regulatory compliance. Once filtration goals are established and the system is operational, Integrity Testing is the most certain way for system operators to know if the filters are working as required. Filtration performance can be affected by filter damage during shipping, improper installation, cleaning chemicals/water temperatures, and process upsets.
When should I integrity test?
Since in-process Integrity Testing is used to make sure your filters will perform as designed, many strategies can be employed. Which strategy is right for your process requires evaluation of your filtration goals and the criticality of filter performance.
- Before installation to ensure there was no damage in shipping and handling
- After installation, make sure the filters are properly installed (no O-ring leaks, etc.) and undamaged
- After in-line sterilization - chemical, hot water, steam - to make sure no damage occurred
- After autoclaving (performed on a filter after removal from its housing) and before re-installation
- Pre-process operation (to make sure the filters are undamaged and properly installed)
- Post-process operation (to make sure no damage or upsets occurred during processing). This is employed in bio/pharmaceutical operations where the sterile product is critical.
How do I test my filters for integrity?
There are several ways filters can be tested for integrity. The most common and effective tests are discussed below. Regardless of which test you use, the cartridge filter must be fully wetted. Here is an example of the pre-wetting procedure.
Forward Flow Diffusion Test
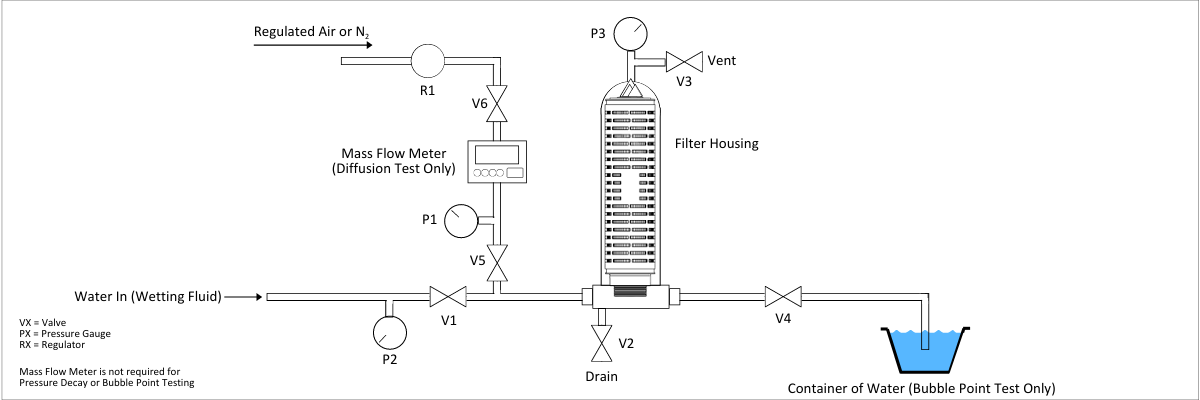
Most filter manufacturers use a Forward-flow Diffusion Test as a final test before product release. This involves making sure the filter is properly wet out (usually with water but could be done with alcohol/water mixtures for hydrophobic filters), then applying a controlled upstream pressure somewhat below the bubble point of the membrane. This allows diffusional flow through the liquid layer but no bulk flow through open pores. This diffusional flow rate is validated against bacteria retention to ensure sterilizing or bioburden reduction performance.
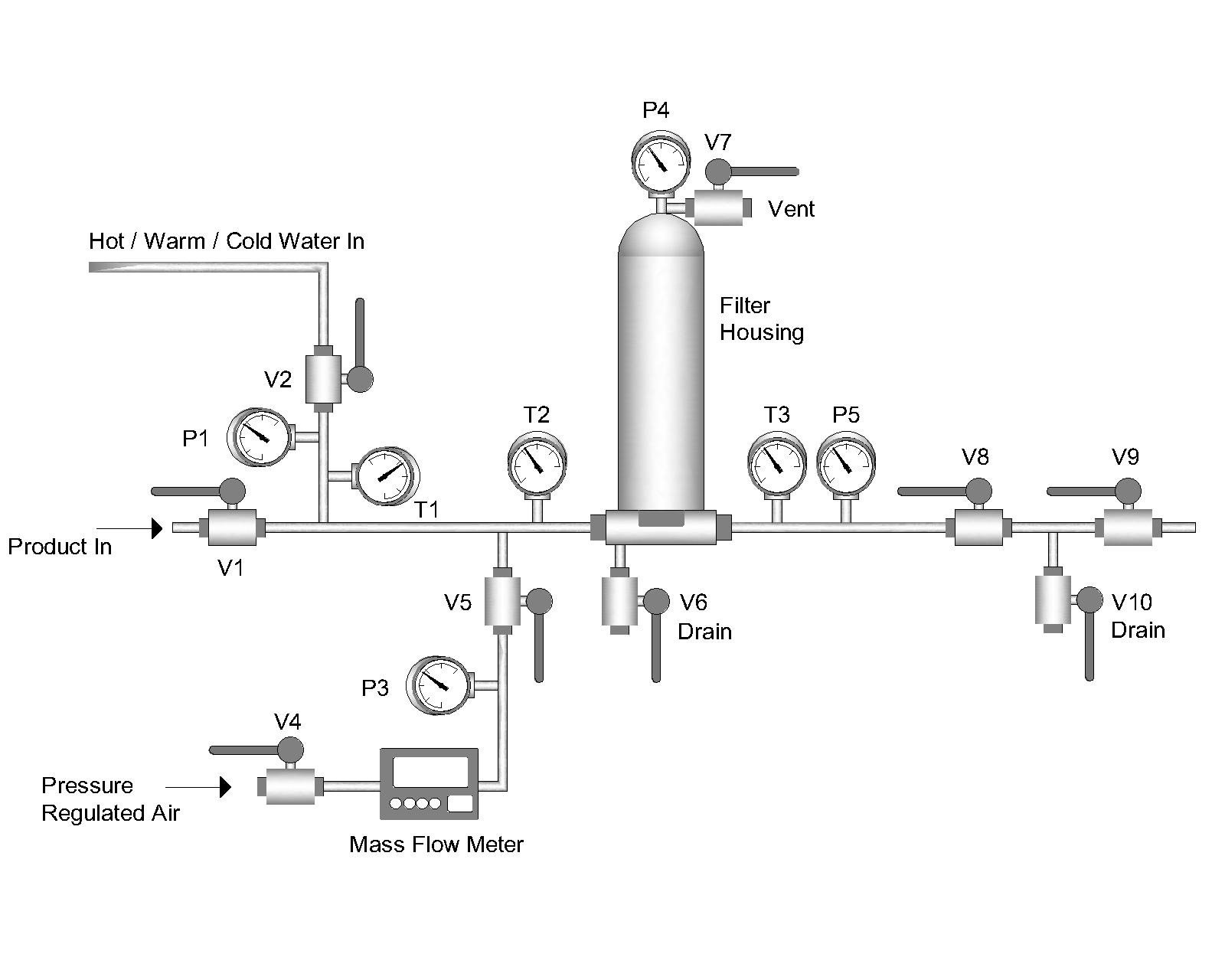
In-process this is accomplished by installing a high-purity pressurized air (or nitrogen) line upstream of the filters to be tested. After installing and flushing the filters, the specified pressure is applied and the flow rate is measured. This can be performed manually by installing a mass flow meter in the airline, allowing the air flow rate to stabilize and recording the value to ensure it meets the filter manufacturer’s specifications. For installations requiring frequent integrity testing (i.e. pre- and post-every batch), an automated integrity test system (available from several suppliers) can be installed.
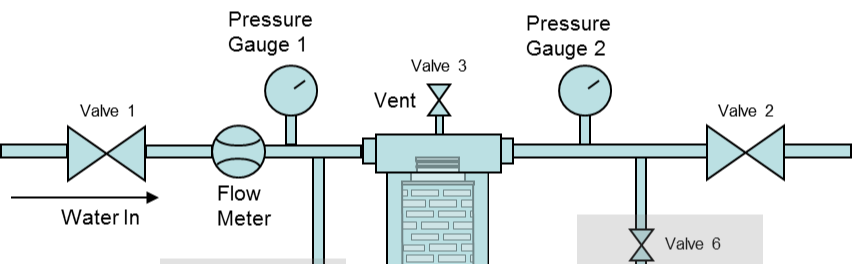
Pressure Decay Test
A simpler integrity test for less critical applications is a Pressure Decay Test. This requires the same air (or nitrogen) source and an accurate pressure gauge but does not require a mass flow meter. To perform this test, after the filters are installed and flushed, a specified pressure is applied to the filters, and the air feed valve is closed. After a defined time interval, the pressure is noted, and the pressure decay is calculated (initial pressure minus final pressure). The allowable pressure decay to guarantee integrity could vary depending on the type and number of filters installed. This test again relies on the diffusional flow allowed at a defined pressure below the bubble point (hence the drop in upstream pressure), and the allowable pressure decay is calculated accordingly. Your filter supplier will supply details on the pressure decay integrity test.
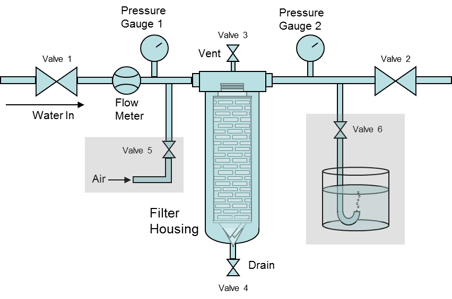
Bubble Point Test
Another option for testing integrity is measuring the bubble point of a filter. This is more typically performed on filters before installation into the system, but proper plumbing could be performed after installation as well. Measuring the bubble point requires installing the filter(s) into a housing, and making sure they are fully wet out with the required fluid (usually water for hydrophilic filters or an alcohol/water mixture for hydrophobic filters). The housing is connected to an upstream air source, and the downstream line is directed into a vessel containing water in which bubbles could be seen. The upstream pressure is slowly increased until a steady stream of bubbles is noted in the downstream vessel. Verifying that this bubble point meets the manufacturer’s specifications indicates that the filter is undamaged and suitable for use.
For more details and instructions on performing the tests described above, see Integrity Testing of Hydrophilic Membrane Filters.
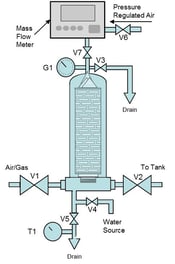 Water Intrusion Test
Water Intrusion Test
For testing hydrophobic filters a water intrusion test can be performed. Water intrusion is used where it is not desirable to introduce alcohol into the system. This involves installing the filter(s) into a housing, applying water pressure on the upstream side at a defined pressure, and measuring any downstream water flow. An integral filter will not allow significant water flow. If the downstream flow exceeds the manufacturer’s specifications it is an indication of damage or incorrect installation. For more details and instructions on performing the water intrusion test, see Water Intrusion Integrity Testing of Hydrophobic Membrane Filters.
Need support with your In-process Integrity Testing?
In-process Integrity Testing is a critical component of assuring final product quality by making certain the filters are operating as needed throughout the process. Doing so lowers risk and the expense associated with disposing of a bad batch.
We're here to help. An evaluation is as simple as a conference call with our Application Engineers to review your current situation and make recommendations on how and where to implement Integrity Testing for your process.
Critical Process Filtration offers over twenty years of experience as a mid-size manufacturer of process filters. We have an experienced Applications and Process Support staff that will identify the right integrity test for your process and will assist with installation, training, and validation as required. For more information on Integrity Testing in your process, contact us here.