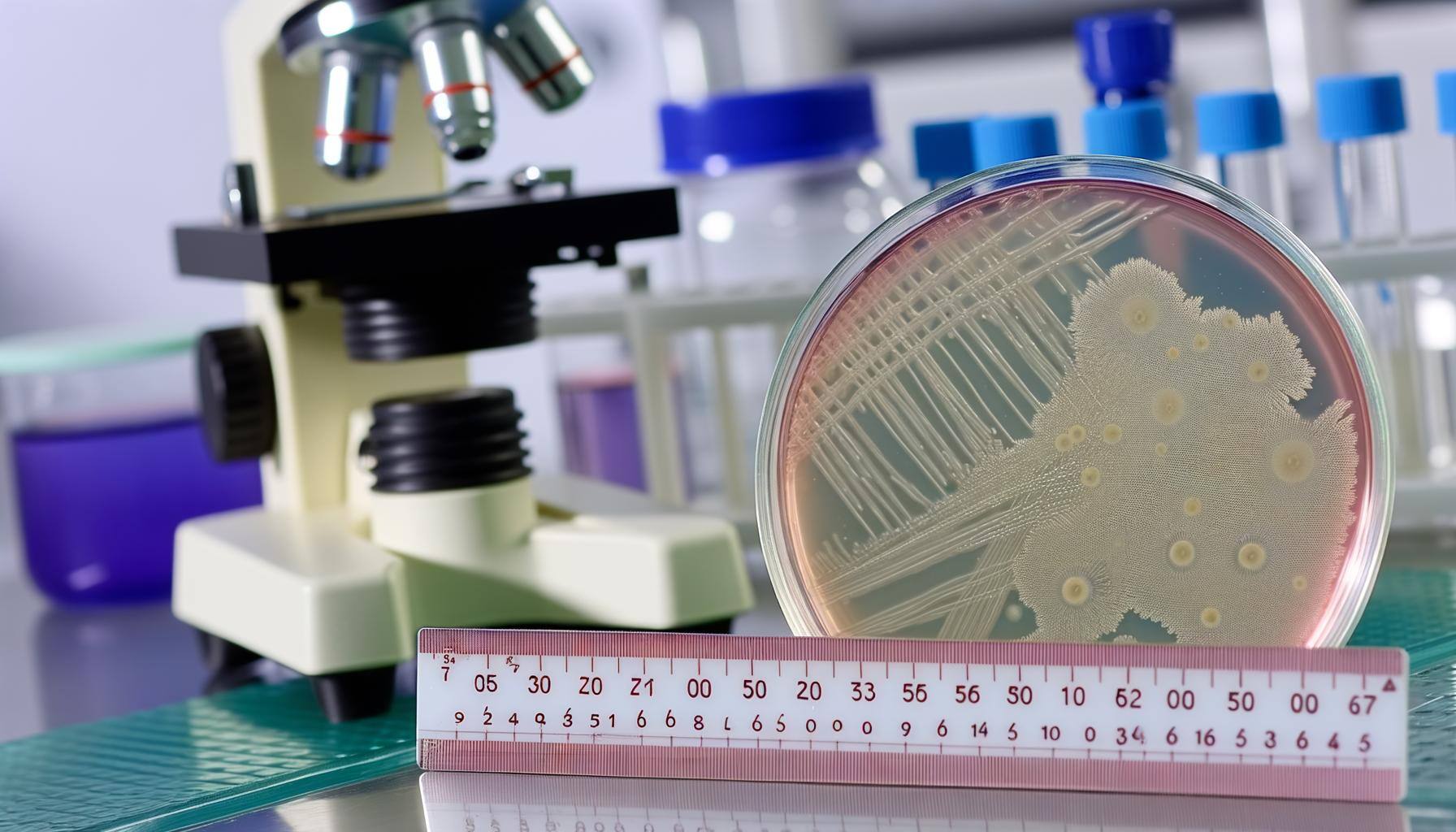
Extractables & Leachables
Extractables and Leachables, these terms are a part of the manufacturing process of new medicines and other areas of such. These terms are very commonly misunderstood by one another, and they both have their distinct characteristics, which can be traced through the whole supply chain.
The definitions of these are not balanced. They overlap and have different utilization, and situations in the manufacturing process must be categorized to highlight the weight and necessity of extractables and leachables.
What are extractables and leachables? Well, the FDA defines them as this:
- Extractables - "These are compounds that can be extracted from a device (in our case a filter) in the presence of a solvent.
- Leachables - "These are compounds that leach into the drug/product formulation from the device (in our case a filter) as a result of direct contact with the formulation.
Compounds Found in Extractables & Leachables:
- Plastic additives, such as stabilizers and processing aids
- Monomers or low molecular weight species
- Manufacturing residues
- Inks and adhesives from secondary packaging
Extractables
Extractables are chemical compounds that may shift from our filters into solvent solutions and predetermined solvent solutions under controlled conditions. These solvents and conditions are selected to vary temperature, pH, polarity, and time. Extractables are designed to obtain a fingerprint of different chemicals that can be extracted using standardized conditions. The toxicological review that happens after the extractables are obtained is crucial for the risk assessment of the product and selecting the right task at hand.
The main purpose of extractable testing is to qualify filter components, screen for and monitor if toxic materials are present, develop analytical methods for leachable testing, and show quality control for acceptance of the filter components.
Here at Critical Process Filtration, we have outsourced valuation testing on our products for extractables testing. This gives the upper hand to our customers when it comes to the leachable testing taking place under the normal conditions use of the filters.
Leachables
Leachables are chemical compounds that may shift from filters into process solutions under standard process conditions. In this process, leachables may end up in the final product. With studies being done on the product for leachables you can assess potential alterations to the product.
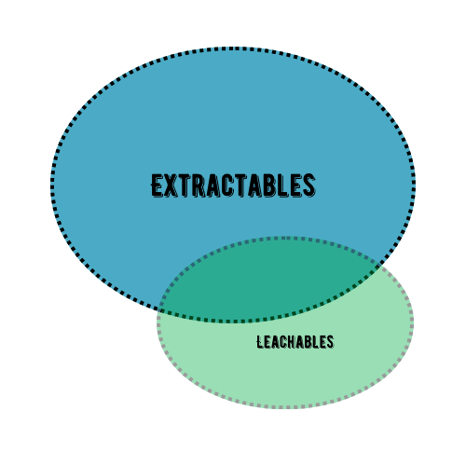
Leachables testing has the purpose of monitoring and control impurities derived from filters in the DP (Drug Products) during stability and/or as part of filter qualification studies. Leachables are a subgroup of extractables and can include many of the same compounds that extractables have. As shown in the figure below.
The Technical Service Team will work with customers to create test plans and conduct tests that simulate actual operating conditions closely. This is done for all application tests, regardless of the industry, but it is a requirement for validating a pharmaceutical process. These tests are carried out at a laboratory scale using flat or encapsulated disc filters.
If you have any more questions about our filters and need help determining the test values or additional information, contact Critical Process Filtration or Call us at (603) 880-4420.
Critical Process Filtration, Inc., an ISO 9001-certified manufacturer of process filters, has been helping customers for over 20 years to build and improve process filtration systems. Proven products, technical services, support, fast shipping, and very competitive pricing assure companies of getting the best value and expertise. Our comprehensive testing, analysis, and validation support your team whenever they need it. Partnering with your process team is how we deliver your company's solutions right the first time.